Test Your Knowledge on Aluminium extraction – mining and electrolysis
Have you just done a science lesson on aluminium extraction – mining and electrolysis? Complete this assessment quiz to test your knowledge!

What form is aluminium present in rocks?

Iron ore occurs naturally as rocks underground in many countries around the world, including the US, Russia, Australia, Brazil, Sweden and Canada. It is mined using open cast mining.
What is the name of the compound produced when bauxite is purified?

The bauxite is purified into aluminium oxide or alumina (a pure white powder) so it is easier to transport to refineries across the world.
What is the name of the process used to extract aluminium from alumina?

Electrolysis is used to extract aluminium from the alumina.
What other elements are needed in the extraction of aluminium from its ore?

How often must the graphite be replaced in the electrolysis reaction?

Graphite anodes and cathodes must be replaced often as the carbon is used up in the process.
Which of the following is NOT a reaction that happens in the electrolysis?
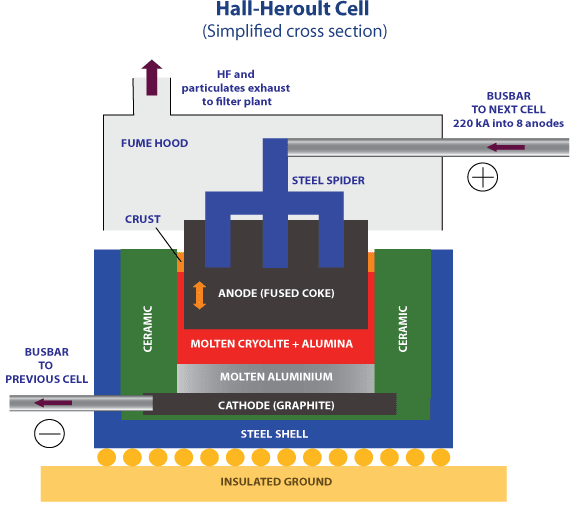
What are the main waste products of the electrolysis process?

Carbon dioxide and oxygen are the main gases emitted from the electrolysis process.
What temperature must the aluminium oxide be heated up to in the electrolysis process?

What process do you think would be better than mining aluminium from the ground and then extracting the alumnium through electrolysis?

Has this lesson made you aware of the energy cost and climate change implications of aluminium production?

You got {number correct}/{number of questions} correct answers
Bad Luck!
You didn't do well this time.
Try again after reading a bit more about aluminium production...

You got {number correct}/{number of questions} correct answers
Well done on a great score!
You definitely have some understanding of aluminium production. Maybe you could aim for even more correct answers next time!

You got {number correct}/{number of questions} correct answers
That's a great score!
You have excellent knowledge of all the facts! People learn from you, but don't forget there is always room for improvement. Keep on learning and inspiring others. The world needs you!
